Cambridge, England
On August 15th, 2016; I visited the University of Cambridge. The University of Cambridge is a collegiate public research university in Cambridge, United Kingdom. Founded in 1209 and granted a royal charter by King Henry III in 1231, Cambridge is the second-oldest university in the English-speaking world and the world’s fourth-oldest surviving university.
The main reason for me be at the University of Cambridge was to visit the scientifically famous Cavendish Lab and its on site Museum. The museum was a self guided tour in a 3 story hallway.

Why would I be interested in this location. Well, the Cavendish Laboratory has an extraordinary history of discovery and innovation in Physics since its opening in 1874 under the direction of James Clerk Maxwell, the University’s first Cavendish Professor of Experimental Physics.

Since its foundation, the Laboratory has had great fortune in appointing Cavendish professors who, between them, have changed completely our understanding of the physical world. Maxwell did not live to see his theories of electricity, magnetism and statistical physics fully confirmed by experiment, but his practical legacy was the design and equipping of the new Laboratory. Maxwell died in 1879 at the early age of 48 and was succeeded by Lord Rayleigh, who was responsible for setting up a systematic course of instruction in experimental physics, which has remained at the core of the Laboratory’s teaching programme.

JJ Thomson succeeded Rayleigh in 1884 and began the revolution in physics which was to lead to the discovery of quantum mechanics in the 1920s.

Among the first generation of physics graduate students were Ernest Rutherford and Charles Wilson, who, along with JJ Thomson, were to win Nobel prizes for their researches. The discovery of the electron by Thomson, the invention of the Cloud chamber by Wilson, the discovery of artificial nuclear fission and the atomic nucleus by Rutherford are examples of the extraordinary advances in experimental technique which ushered in what became known as modern physics.


In 1919, Rutherford announced that, when nitrogen atoms are bombarded by alpha particles, energetic long-range hydrogen ions are produced. The disintegration chamber is a brass tube, (pictured above). The alpha particle source was a radium coated on a disc that could slide along the tube to change the distance from the source to the target. The target material was inserted as a gas or onto a gold foil in front of the source. The hydrogen ions passed through silver foil and were detected on a screen coated with zinc sulphide by the scintillations they produce. The detection of energetic protons indicated that they were the products of nuclear disintegration.

James Chadwick was Rutherford’s second-in-command in Cambridge. Following the observation of neutral particles by the Joliot-Curies, (which they interpreted as gamma rays), he proposed instead that, when alpha particles collide with beryllium nuclei, the neutral ray were in fact newtrons.

Chadwick proved that the emitted particles were indeed a neutron by measuring its mass and momentum. He measure the kinematics of the incoming particles with the devise pictured below.

Lawrence Bragg succeeded Rutherford as Cavendish professor in 1938 and developed the use of X-ray crystallography as an extraordinarily powerful tool for understanding the structure of biological molecules. The culmination of these studies was the determination of the double-helix structure of the DNA molecule by Francis Crick and James Watson.

It is from this era that we have the Thomson’s discovery of the electron, pioneering work in radio, the understanding of isotopes, the laws of X-ray crystallography, the discovery of the neutron – even the splitting of the atom (by artificially accelerated means if you want to split hairs as well as atoms). Perhaps most surprisingly, after World War II the Cavendish turned its attention from the physical to the living world, and the structure of DNA was worked out there by a young American scientist and his British mentor. Their names? James Watson and Francis Crick.
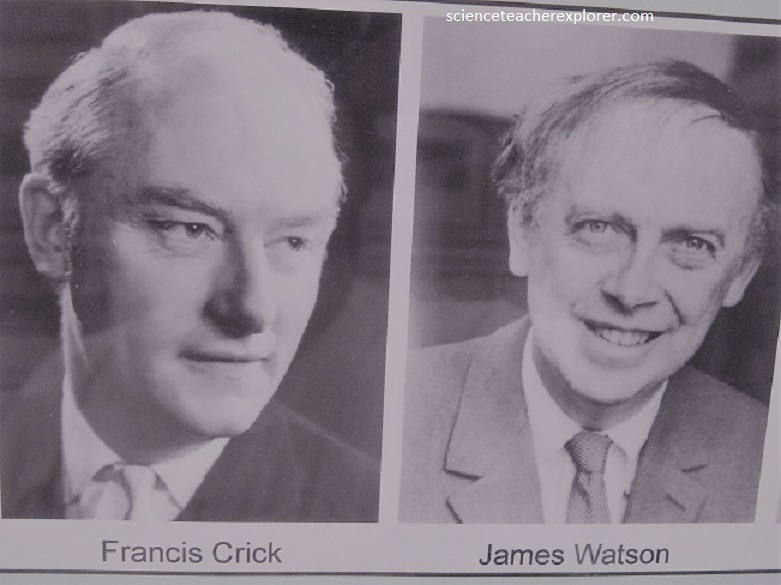

Bragg was succeed by Nevill Mott in 1954 and under his leadership, many pioneering studies were carried out in what is now be termed condensed matter physics, including his own work on amorphous semiconductors which was to lead to his Nobel prize. The Laboratory continued to expand at a great rate until the site in central Cambridge became so overcrowded that a move to a new green-field site in West Cambridge in 1974, (where this lab & museum is now found).

In summary: the following are the best-known Cavendish achievements: J. J. Thomson’s discovery of the electron, Bragg’s law of diffraction, Rutherford’s discovery of the atomic nucleus, James Chadwick’s discovery of the neutron, the deduction of the structure of DNA by Francis Crick and James Watson, the discovery of the pulsar by Jocelyn Bell and Antony Hewish.
